(Note: While the authors stated serum in the publication, the blood samples were collected in EDTA tubes. Thus, in the following summary, serum is corrected to plasma.)
Autologous dendritic cells (DC) as a therapeutic vaccine for cancer was designed to harness natural host immunity to improve cancer treatment. However, DC vaccines have only demonstrated limited clinical success. The variables associated with isolation and differentiation of DCs is just one major factor that contributes to differing efficacy. Additionally, the tumor microenvironment of the patient may be immunosuppressive enough to overcome the DC vaccine. The study’s authors collected plasma prior to treating patients with metastatic melanoma with expanded DCs pulsed with autologous tumor cell lysate. This report examined correlation of plasma immune biomarkers in response to DC vaccine therapy.
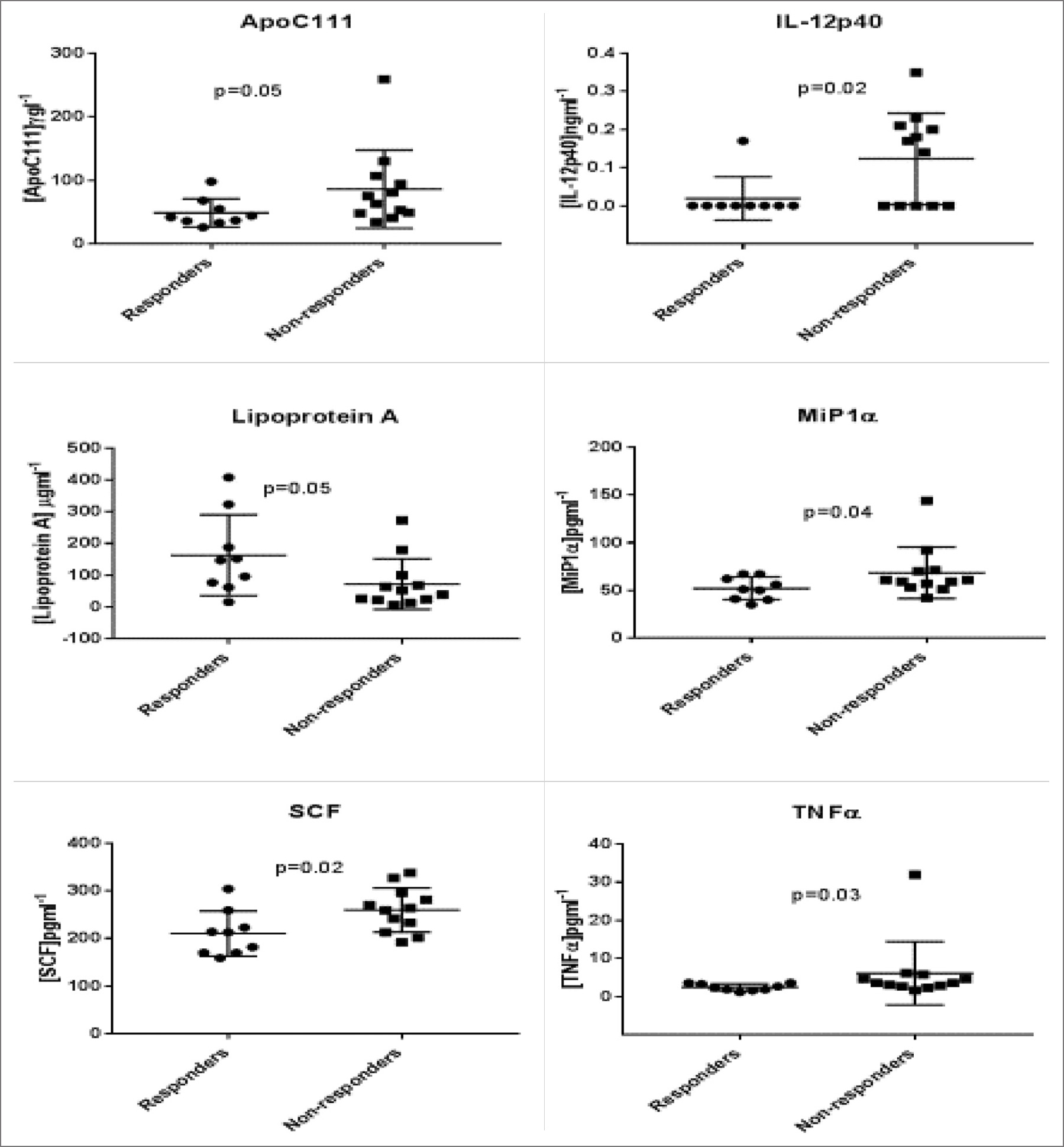
Plasma was collected at 4 and 12 weeks and at 72 hours prior to the first dose of the DC vaccine. Samples were analyzed by Myriad RBM using the human Multi-Analyte Profile (MAP) bead array. There were 12 clinical non-responding patients and 9 responding patients in this study.
There were several plasma proteins that demonstrated significant differences pre-vaccination between responders and non-responders. No differences between the patient groups were observed in plasma proteins at 4 and 12 weeks post-vaccination. These markers indicate that non-responders have a general inflammatory circulating protein profile compared to patients who responded to DC vaccine therapy. It is interesting to note that pre-treatment with anti-inflammatories have a positive impact on subsequent DC vaccine therapy, both in animal models of cancer and in multiple myeloma patients. This report supports the idea that pre-treatment with anti-inflammatory drugs may positively impact cancer patients who receive immune modulatory therapy.